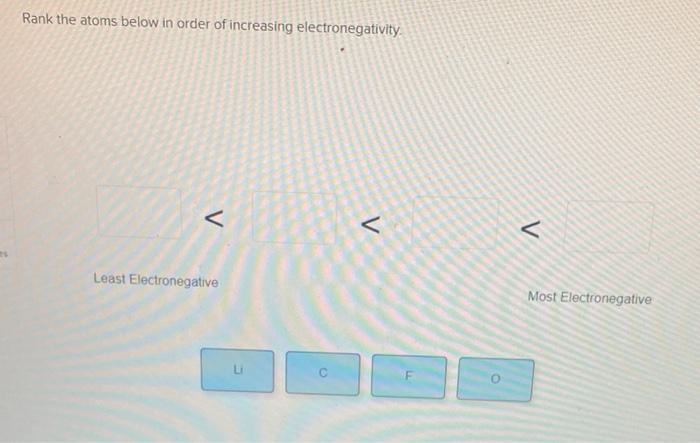
S СІ F N I III II IV У O I II. See the answer Rank the atoms below in order of increasing electronegativity C F O Li Expert Answer 100 4 ratings.
Solved Rank The Atoms Below In Order Of Increasing Chegg Com
Li Na H b.

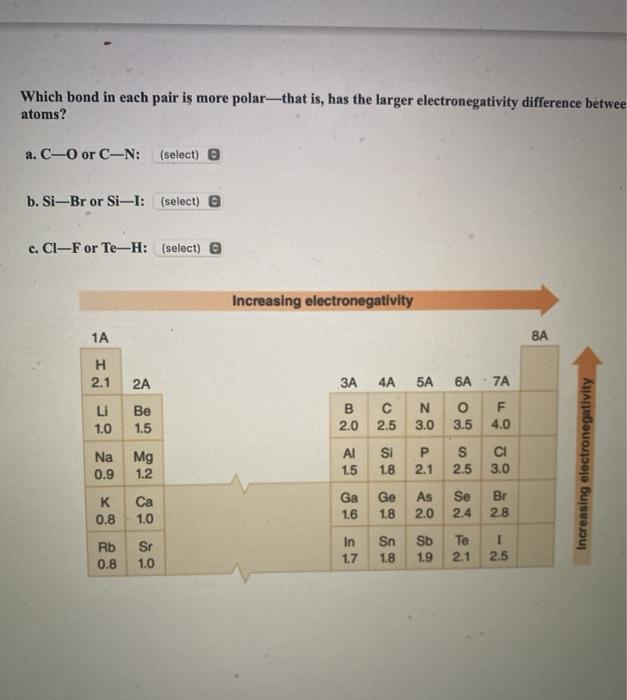
. Whereas when we move across a period then there is decrease in size and increase in electronegativity of an atom. B O N Expert Solution Want to see the full answer. The Lewis structure shown is a complete and valid Lewis structure.
Si P S b. A The oxygen and carbon atoms each have an octet of electrons in their valence shells. Rank the following atoms in order of increasing electronegativity putting the least electronegative first.
Check out a sample QA here See Solution Want to see the full answer. For a schematic overview of the periodic table of elements in chart form. A I B II C III.
Electronegativity decreases as you move down a group in the periodic table eg. Science Chemistry QA Library Using the trends in the periodic table rank the following atoms in order of increasing electronegativity. View the full answer.
Values for electronegativity run from 0 to 4. Therefore out of the given options oxygen will be most electronegative in nature whereas potassium will be the least electronegative in nature. The water absorbs more heat and begins to evaporate as a gas 3.
H - C - H. Rank the following atoms in order of decreasing electronegativity putting the most electronegative first. C The hydrogen atoms have filled valence shells.
CIF Н20 - Label the polar bonds in each molecule and then decide if the molecule is polar or nonpolar. Find step-by-step Chemistry solutions and your answer to the following textbook question. Rank the following atoms in order of increasing electronegativity.
Label the most electronegative and most electropositive atom in each group. Rank the atoms below in order of increasing electronegativity Least Electronegative Most Electronegative C This problem has been solved. S Cl N F I II IV III Ans.
Rank the atoms in descending order of the number of bonds that they commonly form. See the answer Show transcribed image text Expert Answer 100 1 rating The tendency of an atom in a molecule to attract the shared pair of electrons towards itself is known. D Cl K Br.
It can also be used to predict if the resulting molecule will be polar or nonpolar. Thus we can conclude that electronegativity in increasing order will be as follows. Li Be Na Question Rank the atoms in each group in order of increasing electronegativity.
This list contains the 118 elements of chemistry. C III II IV I D I II IV III. P Na Cl c.
An ice cube is dropped onto hot sand which begins to lose energy as the ice cube starts to warm up. Be Mg Ca c. List the elements below in order of increasing electronegativity - Br - Ge - As - K - K - Ge - Br - As F TF.
Check out a sample QA here See Solution. Using the trends in the periodic table rank the following atoms in order of increasing electronegativity. The liquid water in the sand continues to warm up as even more heat from the sand is transfer.
Which of the following molecules contain 16 valence electrons. The electronegativity chart describes how atoms can attract a pair of electrons to itself by looking at the periodic table you can identify and determine electronegativity values of elements from 0 to 4. F Cl Br I each step down a group increases the atomic radii as a new shell of electrons are added and.
See the answer Show transcribed image text Expert Answer 100 3 ratings. Which group of elements is listed in order of increasing electronegativity. Less ------ More Li C O F Sort the elements below by the charge that they will bear as ions.
O C Be c. A value of 40 is assigned to fluorine the most electronegative element. - N2O - CO2 - C2H6 - C3H4 - CH2O - N2O - CO2 - C3H4 C.
118 rows Chemical elements listed by electronegativity The elements of the periodic table sorted by electronegativity click on any elements name for further chemical properties environmental data or health effects. Rank the atoms below in order of increasing electronegativity C F O Li This problem has been solved. D The oxygen atom has four valence electrons that are not being shared with another atom.
The increasing order of electronegativity is given below. Rank the following atoms in order of increasing electronegativity putting the least. Pauling Electronegativity Scale In the following two sets rank the elements in order of increasing electronegativity 1 being the least electronegative.
C N O F as you move left to right nuclear charge increases so there is a greater attraction for electrons. A F Cl Ge Sn b Rb Ca Sc Cs c Zr V Nb Ta d Sn As P S e Li Na K F Chemistry The Periodic Table Periodic Trends in Electronegativity 1 Answer Stefan V. View the full answer.
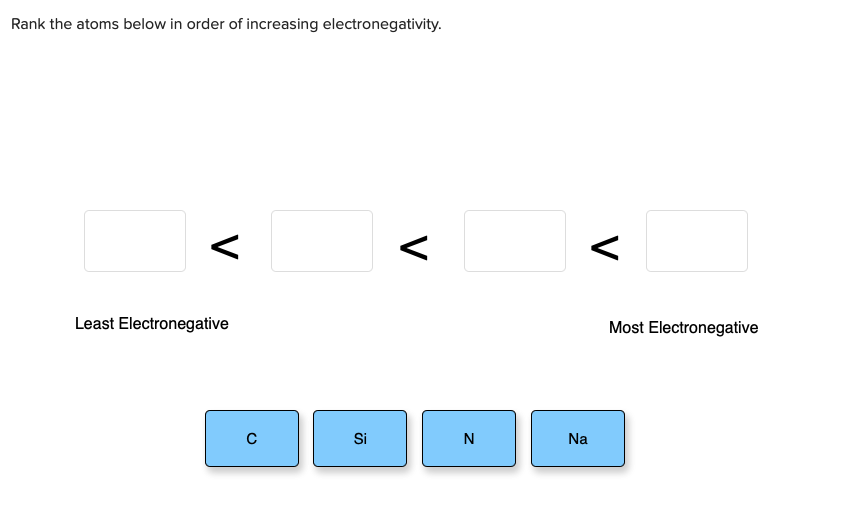
Follow the curved arrows to draw the second resonance structure for the ion below. Least Electronegative Most Electronegative с Si N Na This problem has been solved. Label the most electronegative and most electropositive atom in each group.
Cl S F d. Rank the atoms in each group in order of increasing electronegativity. Be Mg Ca c.
Se Cl Br d. Si P S b. Se O S b.
Chemistry Chemistry questions and answers Rank the atoms below in order of increasing electronegativity C F O Li Question. Rank the atoms below in order of increasing electronegativity. Cl I F d.
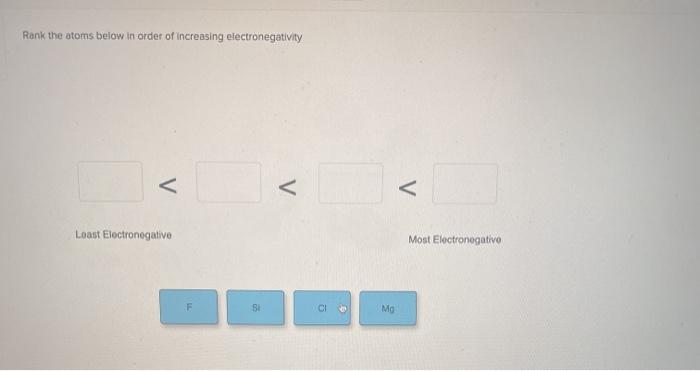
You may want to refer to a periodic table for assistance. Check out a sample QA here See Solution star_border. Rank the atoms below in order of increasing electronegativity Least Electronegative Most Electronegative F Si Mg CI.
Li Be Na Expert Solution Want to see the full answer. As we move across the period electronegativity increases so N is less electronegative. B Two electrons are being shared between the carbon atom and the oxygen atom.
Se Cl Br d. See the answer Show transcribed image text Expert Answer 100 1 rating. 119 rows Electronegativity is a chemical property which describes how well an atom can attract an electron to itself.
Electronegativity is used to predict whether a bond between atoms will be ionic or covalent. As you can see electronegativities generally increase from left to right across a period and decrease down a group. Rank the atoms below in order of increasing electronegativity Least Electronegative Most Electronegative С This problem has been solved.
Electronegativity increases left to right across a row in the periodic table eg. The Periodic Table contains a lot more information than merely the names of each of the chemical elements. Rank the atoms below in order of increasing electronegativity.
Apr 11 2015 I would say d Sn As P S. Sequence the steps below to describe heat flow.
Solved Be Sure To Answer All Parts Rank The Atoms In Each Chegg Com
Solved Rank The Atoms Below In Order Of Increasing Chegg Com
Solved Rank The Atoms Below In Order Of Increasing Chegg Com

Solved Be Sure To Answer All Parts Rank The Atoms In Each Chegg Com
0 Comments